 
Metals higher in the reactivity series (e.g. The Blast Furnace can be used to extract any metal lower than carbon in the reactivity series (e.g. copper or iron.Ĭlick on one of the elements in the reactivity series above to see if its oxide reacts with carbon.Ĭarbon, in the form of coke, is used to extract iron from iron ore (a mixture of iron oxides) by heating them together in a Blast Furnace. Although it doesn't form salts (for instance, you never get carbon sulphate or carbon nitrate), it grabs oxygen atoms from oxides of unreactive metals e.g. Magnesium + water → magnesium oxide + hydrogenĬarbon is also often included in the reactivity series, and I have included it in the series above. Potassium + water → potassium hydroxide + hydrogenĬalcium + water → calcium hydroxide + hydrogen magnesium or zinc) have to be heated with water in order to get them to react: Metals from calcium upwards will react spontaneously with water, but metals lower than that (e.g. Metals higher than hydrogen in the table will also react with water, again producing hydrogen gas. (Sodium and potassium are both lighter than water, so they float) Potassium is even further up, and reacts so violently with acids that the hydrogen produced catches fire! Sodium is near the top of the table, so it reacts more strongly. Zinc, for example, is just above hydrogen, so it bubbles away happily. The further up the reactivity series a metal is, the more vigorously it reacts with acids. Metals below hydrogen in the reactivity series generally don't react with acids, although they will still react with concentrated nitric acid as it is a powerful oxidising agent (but that's a different story. Acids are like salts except they have hydrogen instead of a metal ion. Metals above hydrogen in the Reactivity Series react with acids to produce hydrogen gas. Hydrogen and Carbon in the Reactivity SeriesĪlthough not metals, hydrogen and carbon are often included in the Reactivity Series. Experiments show that Rubidium is indeed more reactive than potassium and Caesium is even more reactive than Rubidium. It follows that elements below potassium in the table should be more reactive than potassium is. Similarly, atoms of potassium ( K in the table) are larger, and so more reactive, than atoms of sodium. The further down a group an atom is, the larger the atom is, and the more reactive the element is.Ītoms of sodium ( Na in the table) are larger, and so more reactive, than atoms of lithium ( Li in the table). This means that the nucleus exerts less of an electrostatic pull on the outer electron. 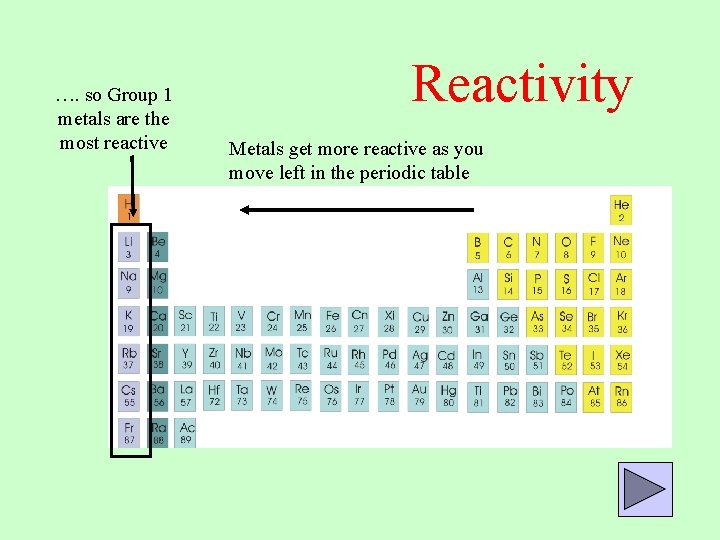
The reactivity of metals increases as you go down the group because the outer electrons (which are lost when the metal reacts) are further from the nucleus. Losing each electron requires some energy to be put in, so it is easier for sodium to react than magnesium, and easier for magnesium to react than aluminium. Aluminium reacts by losing three electrons. Magnesium reacts by losing two electrons. 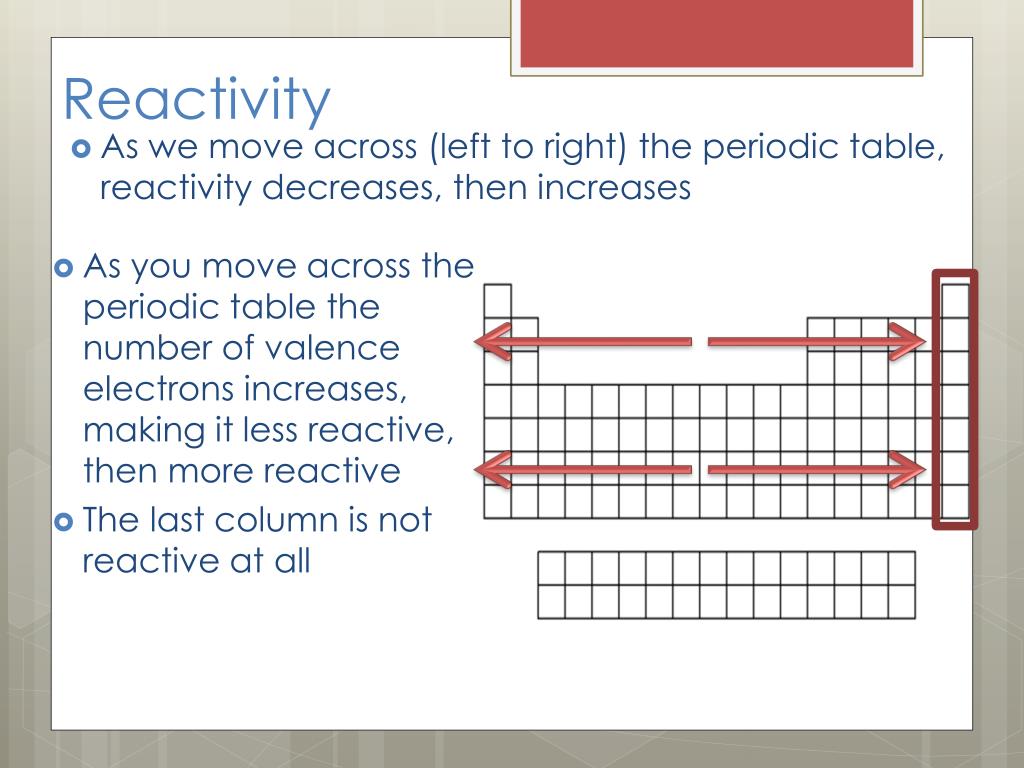
Sodium reacts by losing a single electron. Energy is needed to strip those negative electrons away from the positive nucleus - since they are opposite in charge, they are attracted to each other - and the more electrons a metal atom has in its outer shell, the harder it is to get that atom to react. The second equation shows us that tin is more active then copper.The reactivity of the metals decreases as you move right across the table because the atoms have more electrons that they need to lose. In the first reaction we determined that zinc is more active than copper and the third reaction shows us that zinc is also more active than tin. Using all three of the reactions above, we can make an activity series for Cu, Zn, and Tin. If a piece of zinc is placed in the SnCl 2 a reaction occurs. In this case the tin is more active than the copper, as the copper is not able to force its electrons onto the tin. Suppose a piece of copper was placed in a solution of SnCl 2, and nothing happens. When an ion keeps the same charge in a reaction and is just there to balance out the opposite charges, we call it a Spectator Ion. Nitrate ion in both the reactants and the products has a -1 charge. In the equation below, nitrate is there to balance the charges. Positive ions (cations) can't just be floating around by themselves in solution, they have to have a negative ion (anion) to balance them. We say that Zn is more active than the copper. \): An activity series for common metals.Įxample: Zn and Cu both tend to lose 2 electrons, but the zinc wants to lose them more, so much so that it can force the two electrons on Cu 2 + and make it become Cu again. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
